English | Spanish
Dynic Global 800-326-1249
English | Spanish
Dynic Global 800-326-1249
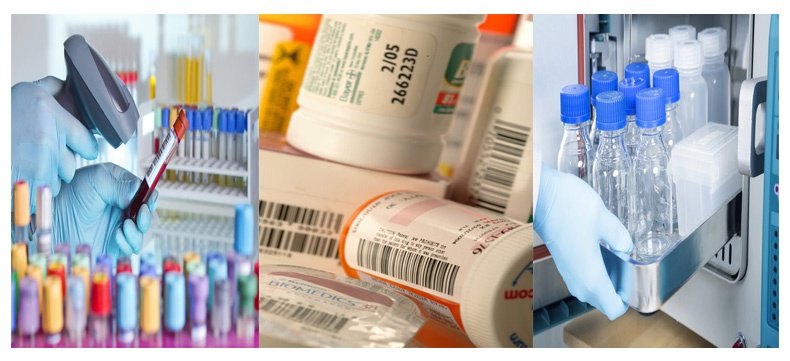
Where bar codes and expiration dates are critical
Health & Pharmaceutical applications where Dynic products can be found include patient identification, lab samples, asset tracking, and package labeling on medicines. It's critical that barcodes in this arena withstand common disinfecting solutions such as IPA (isopropyl alcohol) and sterialization proccesses like auto-claving, and remain clearly legible. Our list of TTR that are FDA-listed is extensive.
More information about our getter products is on our HG Sheet page.
If you're a current Dynic customer and need compliance documentation to support your use of our products in these applications, you can login on our Compliance page.
Tel: 800-326-1249 or 503-693-1070
Fax: 503-648-1185
4750 NE Dawson Creek Dr
Hillsboro, OR 97124
Join our community to stay up to date with tips & tricks for excellence in execution.
© 2026 Dynic USA Corporation. All Rights Reserved. | Terms & Conditions | Privacy Policy
S2®, Sirius®, and Cetus® are all registered trademarks of Dynic USA Corporation.